The European Union’s Medical Device Regulation (MDR) has fundamentally reshaped the regulatory landscape, replacing the older Medical Device Directives (MDD/AIMDD). For medical device manufacturers targeting the EU market, MDR compliance is no longer just about meeting requirements—it’s about demonstrating ongoing safety and performance throughout the entire device lifecycle. As an ISO 13485 certified manufacturer with active MDR experience, we provide this practical guide to help brands navigate this complex transition.
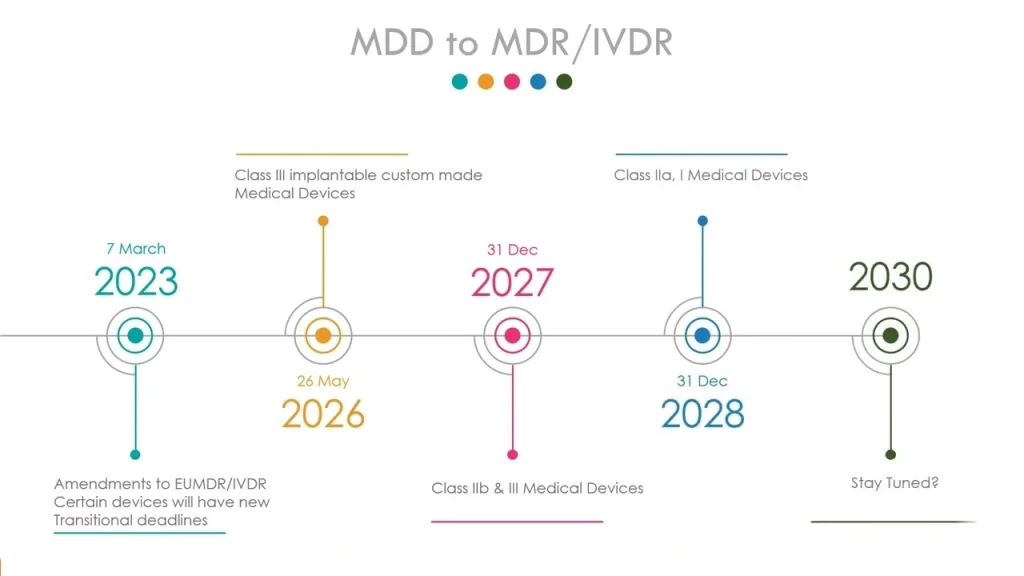
The MDR Paradigm Shift: What’s Truly Changed?
The MDR represents more than an update; it’s a complete philosophical shift in regulatory approach:
- Enhanced Clinical Evidence: Requirements for clinical data are significantly more stringent, even for well-established devices.
- Strengthened Post-Market Surveillance: Proactive and continuous monitoring of device performance is now mandatory.
- Stricter Notified Body Oversight: Notified bodies face more rigorous requirements, leading to more thorough audits.
- Expanded Scope: The regulation now covers certain devices that were previously unregulated or under-regulated.
The Pillars of MDR Compliance: A Manufacturing Perspective
Achieving and maintaining MDR compliance rests on several key pillars that directly impact manufacturing:
1. Technical Documentation & State of the Art
The technical file has evolved into comprehensive technical documentation that must demonstrate the device represents the “state of the art.” This includes:
- Detailed device description and specifications
- Risk management file
- Verification and validation data
- Clinical evaluation report (CER) with post-market clinical follow-up plan
2. Quality Management System Integration
An ISO 13485 certified QMS provides the fundamental framework for MDR compliance. Specific MDR requirements integrated into the QMS include:
- Unique Device Identification (UDI) implementation
- Supply chain control and supplier management
- Post-market surveillance system
- Serious incident reporting and field safety corrective actions
3. Economic Operator Responsibilities
The MDR clearly defines roles and responsibilities across the supply chain:
- Manufacturers bear ultimate responsibility for device safety and performance
- Authorized Representatives must have continuous access to technical documentation
- Importers and Distributors have verification and traceability obligations
How an Experienced Manufacturing Partner Simplifies MDR Compliance
Partnering with a manufacturer well-versed in MDR requirements provides significant advantages:
- Robust Technical Documentation Support: We maintain comprehensive technical documentation for our device platforms, providing a solid foundation for your CER and ensuring alignment with state of the art.
- Integrated Quality Processes: Our ISO 13485 certified QMS already incorporates MDR-mandated processes like UDI, post-market surveillance, and supply chain controls.
- Clinical Evidence Strategy: We help develop appropriate clinical evaluation strategies, leveraging existing clinical data and establishing PMCF plans.
- Notified Body Readiness: Our experience with MDR audits ensures we’re prepared for rigorous notified body assessments.
Practical Steps Toward MDR Compliance
- Gap Analysis: Conduct a thorough assessment of current documentation and processes against MDR requirements
- Clinical Strategy: Develop a comprehensive clinical evaluation plan addressing the new requirements
- Quality System Update: Ensure your QMS—or your manufacturer’s—fully addresses MDR-specific requirements
- Partner Selection: Choose manufacturing and regulatory partners with proven MDR experience
Conclusion: Beyond Compliance to Competitive Advantage
MDR compliance, while challenging, represents an opportunity to demonstrate your commitment to device safety and quality. By partnering with a manufacturer that has embraced the MDR framework, you not only simplify your path to market but also build a foundation of quality that differentiates your products in the marketplace.
Navigate MDR compliance with a partner who speaks the language. Contact Dinghmed to learn how our MDR-ready manufacturing approach and regulatory expertise can streamline your access to the EU market.
